AST to improve coccidiosis management
Poultry producers looking for a way to improve their coccidiosis management strategy should seriously consider anticoccidial sensitivity testing (AST). This is the advice of Dr Marco Quiroz, director of veterinary technical services, global poultry, Zoetis.
By Diana Delmar
“For effective, long-term coccidiosis management (AST), we need to combat resistance and preserve anticoccidial efficacy,” says Dr Quiroz. “Sensitivity testing can help to achieve that goal, but isn’t as widely used as it could be.” AST is, however, just part of a puzzle, Quiroz continues. “The results must be considered along with routine necropsy findings, performance data and the history of anticoccidials used on a given farm. This individualised information can then be used to guide development of an anticoccidial rotation programme that is likely to yield the best results.”
AST procedure
To perform AST, fresh faecal samples are collected and sent to a lab. Eimeria oocysts are isolated, propagated, identified microscopically and are then inoculated into chickens at doses high enough to cause coccidiosis. The inoculated chickens are treated with various anticoccidials and evaluated based on lesion scoring, bodyweight gain and feed conversion. The results are compared to those from inoculated but untreated chickens, and reveal which anticoccidials are likely to be effective against the various species of Eimeria isolated on the farm, Quiroz explains.
Broad-scale sensitivity studies confirm that certain anticoccidials are more effective against different Eimeria strains than others. In one Zoetis-sponsored study, field isolates were collected between 2000 and 2012 from farms in Europe, Africa and the Middle East.
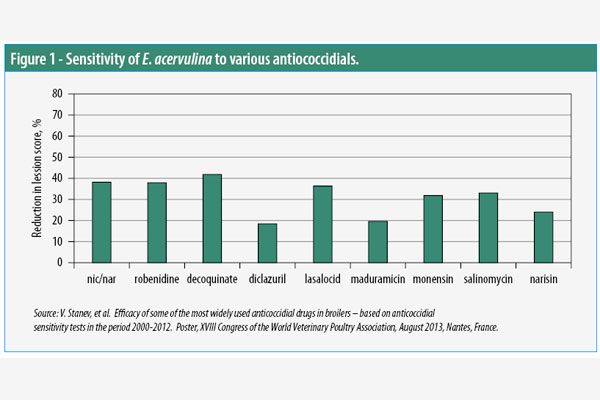
AST was performed at the French National Institute for Agricultural Research. Just one example of the findings based on lesion scoring alone demonstrated that Eimeria acervulina was most sensitive to decoquinate (Figure 1).
Strains of E. maxima were most sensitive to lasalocid, and E. tenella strains were most sensitive to decoquinate, which reduced the lesion score by more than 73%.
Other anticoccidials showed lower efficacy, demonstrated by weaker sensitivity of different Eimeria species. “However,” Quiroz says, “the results could differ on individual farms, depending on which anticoccidials have been used and for how long.”
Restoring sensitivity
If AST indicates widespread anticoccidial resistance, producers may want to consider incorporating coccidiosis vaccination into their programme. Vaccination reseeds the poultry house with Eimeria oocysts that are still sensitive to in-feed anticoccidials.
“AST can play an important role here too by indicating whether vaccination has restored sensitivity to various in-feed anticoccidials,” he notes.
“Although AST can be costly, some animal health companies provide it as a service to customers in some areas,” he says. AST also takes time to perform, but Quiroz believes it is worth the effort because the information provided is valuable.
AST can not only contribute to development of a successful anticoccidial rotation plan, it helps producers avoid wasting time and money on in-feed anticoccidials that aren’t likely to work well, and in this way, combats the problem of resistance.
“Preventing resistance also requires rotating anticoccidials from different classes. If resistance builds against salinomyin, for example, there is likely to be resistance against monensin, since it’s in the same drug class.”
“Be sure to plan ahead by one or two years. It takes time to rotate different anticoccidials through and give each one an adequate rest,” Quiroz concludes.
[Source: World Poultry – Managing Coccidiosis, 2014]
Join 31,000+ subscribers
Subscribe to our newsletter to stay updated about all the need-to-know content in the poultry sector, three times a week. Beheer
Beheer








 WP Admin
WP Admin  Bewerk bericht
Bewerk bericht