Understanding the dynamics of worm infestation in poultry

Worms are still a big problem worldwide and cause decreased production and fertility in layers and broiler breeders. Worms seldom cause issues in broilers, but that could change in the near future. With changing housing systems and a longer lifespan, broilers may be affected too.
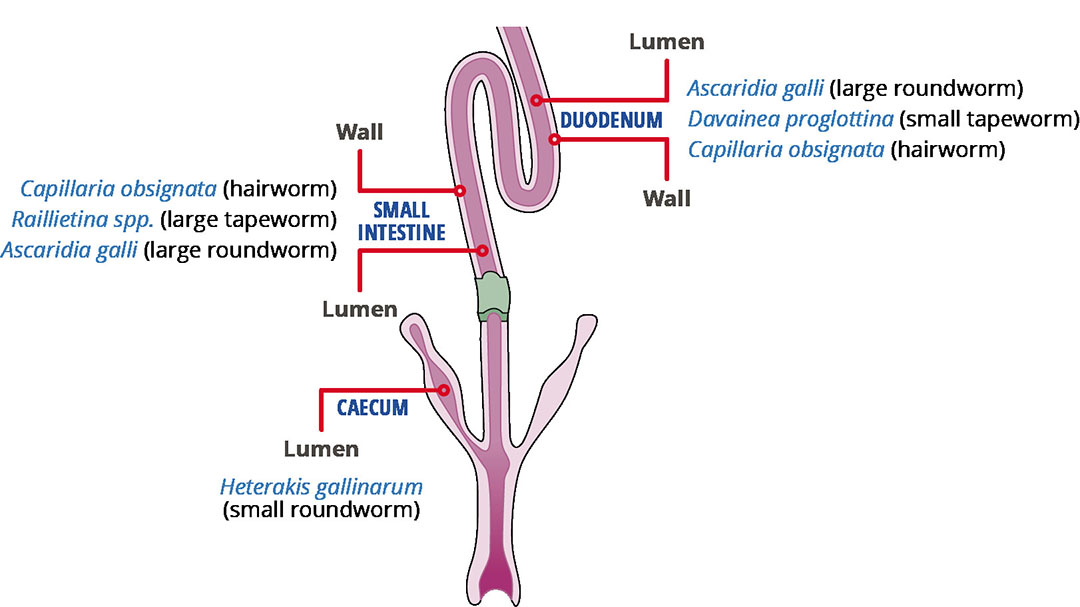
The most common worms found in poultry are members of two taxonomic classes. The first class is nematodes which are the most important roundworms and include Ascaridia galli (large roundworm), Heterakis gallinarum (ceacal roundworm) and Capillaria spp. (hair worms). The second class is Cestoda. These are the major tapeworms and include Raillietina spp. (large tapeworms) and Davainea spp. (small tapeworms). The tapeworm Davainea spp. colonizes the duodenum while small roundworms (Heterakis gallinarum) primarily colonize the caecum (Figure 1). Hairworms (Capillaria), large tapeworms (Raillietina) and large roundworms (Ascaridia) all colonize the small intestines.
Figure 1 – Where parasitic worms colonize the intestinal tract of chickens.
Tapeworms and hairworms cause direct damage to the intestinal wall. The large roundworm causes catarrhal enteritis, with diarrhoea being the primary symptom. The small roundworms cause little direct damage. All worms compete with the chicken host for nutrients through absorption. In parent stock the first signs of infection will be lower egg production, lower feed intake and lower fertility.
Life cycle and prepatent period
Ascaridia galli, Heterakis gallinarum and Capillaria obsignata have direct life cycles. The adult worm produces eggs that are excreted into the environment. Under favourable conditions, such as high humidity and temperature, the eggs embryonate to an infective stage. This embryonating period varies depending on the species. When an infective egg is taken up by the host, it develops into an adult worm. The total time from egg, to maturity, to adult worm, to the first egg, is called the prepatent period. Awareness of this period is very important to be able to devise a preventive strategy. The length of the prepatent period depends on many factors, including environmental conditions (i.e. humidity, temperature), the availability of hosts and chemical challenges (i.e. sanitizers, parasiticides). The prepatent period is always denoted as the shortest possible time based on optimum reproduction.
Histomoniasis
Histomoniasis infection is becoming more common due to the introduction of more ecological husbandry systems and the recent ban on effective drugs in several countries. The life cycle of this protozoal infection is direct, although it uses a carrier (protector) outside the host. The adult protozoa live in the ceca and can also migrate to the liver, causing ulceration in the ceca and necrotic foci in the liver. Outside the host the parasite dies very quickly. The parasitic nematode Heterakis gallinarum acts as a carrier of Histomonas eggs, and earthworms can act as collectors and distributors of the eggs. Histomonas is eaten by Heterakis worms and is incorporated into the Heterakis egg and when shed, the earthworm collects infected eggs. Since Heterakis eggs are very resistant to most disinfectants, it can be difficult to clear an infected farm.
Metronidazole products are the most commonly used drugs to destroy protozoa. Chickens do not acquire immunity against Histomonas and, therefore, prevention must be based on elimination of the intermediate host: Heterakis gallinarum during the course of its prepatent period (24-30 days). In older chickens, the prepatent period tends to be long. In young chickens, de-worming should take place every 4 weeks, and after 12 weeks of age, every 5-6 weeks to break the life cycle of the Heterakis, helping to control Histomonas.
Diagnosis of worm infections
In general, it is recommended to incorporate a worm check in the routine health programme. Critical moments are transfer from rearing to production, the first weeks after production starts and at around 40 weeks of age. Postmortem examinations should be done on healthy flock members with normal feed intake. Infected birds that have not eaten for several days will most likely excrete a lot of adult worms, complicating diagnosis.
As part of the exam, include a microscopic check of the small intestine and cecal content to check for worms and their eggs, as well as coccidia. Check the duodenum, jejunum and ileum of the small intestines and collect a surface sample with content from the ceca. For Heterakis, cut off the ceca top and squeeze a small amount of the content onto a slide. Place a second slide on top of the material and spread it to create a thin layer. Adult Heterakis worms (if present) should be visible with the naked eye. In extreme cases, especially in the case of severe Ascaridia infection, big roundworms in the faeces are a positive diagnosis. However, worms are not always present in the excreta, even when a bird is infected.
Laboratory faecal examination at specific ages is often carried out as a routine part of the biosecurity control system. The frequency of this routine examination must be based on the prepatent period (from egg to larvae, to adult, to new egg) of infections within a region. The number of droppings taken varies with the flock size but, in general, 40 droppings and 10 cecal droppings per house will be sufficient. Mix the droppings into two samples. Centrifugation and flotation methods with solvents for specific gravity are most commonly used. Some solvents may cause eggs from trematoda to become misshapen and, therefore, harder to identify. Common solvents are either NaCl solutions with a specific gravity of 1.19 at 20°C or MgSO4 solutions with a specific gravity of 1.28 at 20°C.
Therapy
Therapy must be divided into curative and preventive: the first to treat the infection and the second to prevent reinfection. A curative treatment should always be followed by a preventive strategy. Check with the manufacturer before treating breeders for parasites during the production cycle, as significant drops in production may occur. Some soluble products may alter the flavour of the drinking water, making it bitter. The altered flavour can reduce water intake and cause production losses. Each facility should develop its own preventive strategy based on local conditions and the available medication. It is important to note that eggs in the litter and floor can never be killed or removed unless the litter is removed and the floor is treated.
A good time for prophylactic treatment is just before birds are transferred from rearing farms to production houses. The treatment should be done at the rearing farm during the period when juvenile worms become adults and thus can be treated. Worms using an intermediate host or a carrier can also be treated by eliminating the host or carrier. With Histomonas, the worm can be the carrier and must be eliminated.
Table – Life cycle characteristics of nematodes and cestodes important to
poultry production
**The two tapeworms Raillietina and Davainea have an indirect life cycle where the egg develops into an infectious cysticercoid in an intermediate host. For Raillietina, flies and ants are known intermediate hosts, while Davainea uses a snail as an intermediate host.
Join 31,000+ subscribers
Subscribe to our newsletter to stay updated about all the need-to-know content in the poultry sector, three times a week. Beheer
Beheer











 WP Admin
WP Admin  Bewerk bericht
Bewerk bericht