Are we underestimating mycotoxin risk?

Alltech’s mycotoxin team has been comparing risk analyses from different mycotoxin testing methods to better understand whether feed and livestock producers may be underestimating mycotoxin risk if relying solely on rapid tests as a detection tool.
MYCOTOXIN SPECIAL 2023 – read all articles
To assess mycotoxin risk, mycotoxin type and concentration must first be determined. Mycotoxin testing types are generally classified into either rapid test methods or the more advanced laboratory-based detection. The primary rapid test methods include lateral flow devices (LFDs) and enzyme-linked immunosorbent assays (ELISA). Of the lab-based methods used for mycotoxin analysis, high-performance liquid chromatography, and ultra performance liquid chromatography with tandem mass spectrometry (UPLCMS/MS) are 2 of the most recognised.
Method influences risk assessment
As a greater number of mycotoxin types are analysed, a better picture of total mycotoxin risk can be gained. But what happens if only a limited number of mycotoxins are tested for? Traditional mycotoxin guidelines that only look at one mycotoxin at a time are not always helpful in understanding the total risk exposure to the animal. This lack of information on the total risk is why the Alltech Mycotoxin Management team developed a tailored risk assessment method back in 2012. Known as Risk Equivalent Quantity (REQ), this method provides feed and livestock producers with a unique way to identify the risk associated with the presence of multiple mycotoxins in raw materials or finished feeds. Using this REQ value to compare the difference between assessing 1 mycotoxin, 6 mycotoxins, and 54 mycotoxins, it is possible to calculate how much risk could be underestimated if only a limited number of mycotoxins were analysed.
Corn grain and sows
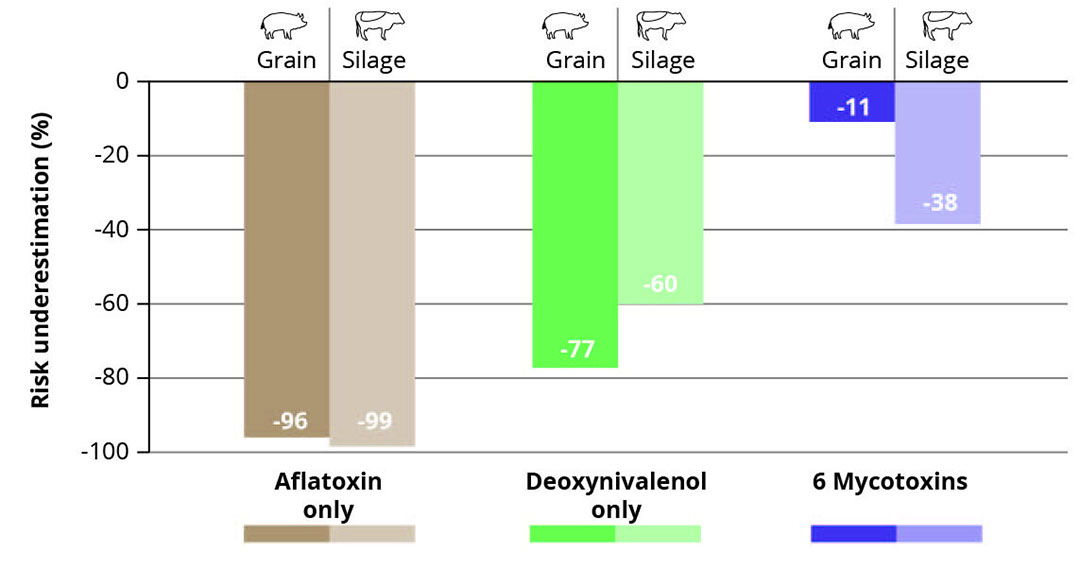
Based on samples analysed by Alltech 37+ (Table 1), if only aflatoxin (AF) was measured, then risk to sows would be underestimated by 96% (Figure 1). This makes sense, as aflatoxins were only detected in 9% of samples whereas other mycotoxins are detected more frequently. In situations where the only option is to test for one mycotoxin, analysing samples for deoxynivalenol (DON) is slightly better than analysing for AF, with an underestimation of 77% of risk, but there are many mycotoxins still not accounted for which are typically present in corn. When corn grain is analysed for all 6 mycotoxins that can be detected through a rapid test method, the overall risk assessment can be closer to what would be delivered by testing for an increased number of mycotoxins. The presence of fusaric acid and other emerging mycotoxins would not be detected by the rapid test methods, but many of the mycotoxins posing the greatest risk to pigs, such as DON, zearalenone and T-2/HT-2, would be included.
Figure 1 – Differences in risk assessment in corn grain and corn silage (sows and dairy cows).
Corn silage and dairy cows
Similar to grain, total mycotoxin risk assessment of corn silage can vary depending on the mycotoxins analysed (Figure 1). In this case, assessment for only AF could result in a 99% underestimation of total risk for dairy cows, as AF was detected in only 9% of samples, with the majority of these containing very low concentrations (median <1 ppb). Sample analysis for only DON would be better — in this case, a 60% underestimation of total risk — but again, many key mycotoxin groups would not be detected. Assessment for 6 mycotoxins comes closer to the total risk assessed by the more advanced laboratory method, but this assessment in corn silage does not get as close to those results as the similar assessment in corn grain. This may be due to the higher occurrence and concentrations of mycotoxins such as fusaric acid, Penicillium mycotoxins and Aspergillus mycotoxins in corn silage.
Using readily available data, the comparisons in these examples were made using Alltech 37+ as the most comprehensive risk assessment method. This method identifies 54 individual mycotoxins. If other LMCS methods were used to identify an even greater number of mycotoxin types, the potential risk underestimation when only relying on rapid test methods would likely be amplified
Mycotoxin testing plan
Although assessment of a limited number of mycotoxins by simpler analytical methods may not provide the full picture of animal’s exposure to mycotoxins, these rapid test methods are still a valuable and accurate means of testing for mycotoxins. Many of these rapid test kits have been validated, with validation results published in scientific journals. The best mycotoxin management program is typically based on a combination of on-site rapid testing and laboratory-based analysis. Rapid tests can be used for routine testing where speed and cost are of concern, but periodic mycotoxin testing by laboratory-based methods can be used to learn about more complex mycotoxin mixtures.
Turning analysis into a control programme
It is important to remember that at any point in time, results of the average mycotoxin analysis can only show you a best-case scenario. At best, mycotoxin levels will remain the same in the tested ingredient or ration, but it is also known that they can continue to increase over time during storage. Equally, the total risk can be greater when multiple ingredient sources are brought together. Without knowing the full mycotoxin profile of a commodity or ration, developing a mycotoxin management programme may be difficult. We also learned from the examples shown above that analysing samples for only one mycotoxin can lead to significant underestimation of total risk, even if the results for that mycotoxin type are accurate. As a greater number of mycotoxins are tested, a more accurate picture of risk can be formed.
Determining the presence of multiple mycotoxins is important, but there may be a limit on the number of mycotoxins assessed. Over 500 mycotoxin compounds have been identified, but it may not be possible to routinely test for this number. Furthermore, for many mycotoxins, it is still unknown how they impact animal performance and health. However, most mycotoxins are detrimental to achieving optimal animal performance, welfare and profitability. To effectively combat mycotoxin risk, a complete mycotoxin management programme is needed, one that focuses on identifying risk, quantifying risk and mitigating risk.
References are available on request.
 Beheer
Beheer






 WP Admin
WP Admin  Bewerk bericht
Bewerk bericht